Lumbar artificial disc replacement surgery may be recommended to treat chronic, severe low back pain caused by degenerative disc disease. Disc replacement is only advised if a minimum of 6 months of nonsurgical treatments are ineffective and if the pain is limiting ability to function in everyday life.
This surgery consists of replacing the painful spinal disc with a device designed to mimic the disc’s natural movement. The goal of artificial disc replacement is to alleviate pain by reducing aggravating micro-motion, stabilizing the spinal segment, and minimizing inflammation.
See Causes of Degenerative Disc Disease Pain
Lumbar artificial disc replacement is a relatively newer procedure compared to lumbar fusion, but has been available in the U.S. since 2000, when FDA studies began. Three lumbar disc devices have been FDA-approved for use in the US, and two are still available.
In This Article:
- Lumbar Artificial Disc Replacement for Chronic Back Pain
- Pain Generated by the Spinal Disc
- Total Disc Replacement – Lumbar Artificial Disc Replacement
- Lumbar Artificial Disc Surgery Recovery
Artificial Disc Replacement and Spinal Fusion
There are two main surgeries designed to address painful and functionally disabling lumbar degenerative disc disease: lumbar fusion and lumbar artificial disc replacement.
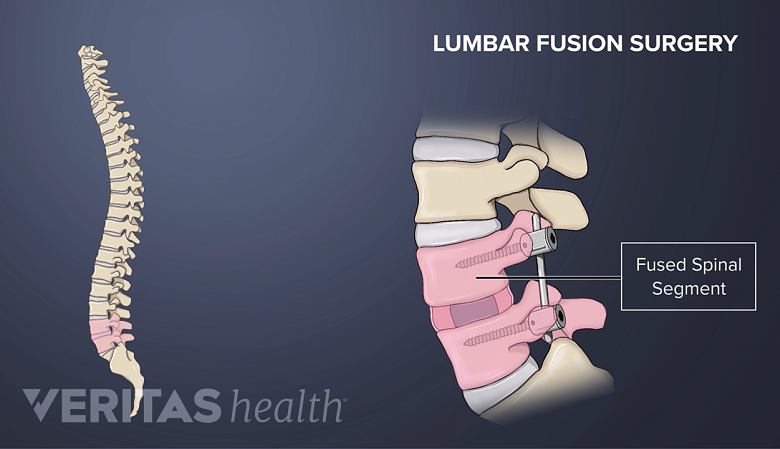
Spinal fusion
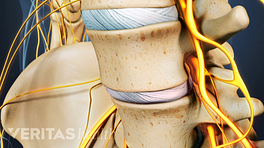
Spinal fusion aims to reduce lower back pain by eliminating movement between vertebrae.
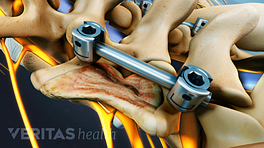
Spinal fusion is the traditional surgery for degenerative disc disease. In this procedure, a disc is removed from the spine and the lumbar segment is immobilized to allow the two adjacent vertebrae to fuse together using a biologic and structural implant such as screws, rods, or plates. The goal of spinal fusion is to reduce lower back pain by eliminating painful movement between two vertebrae.
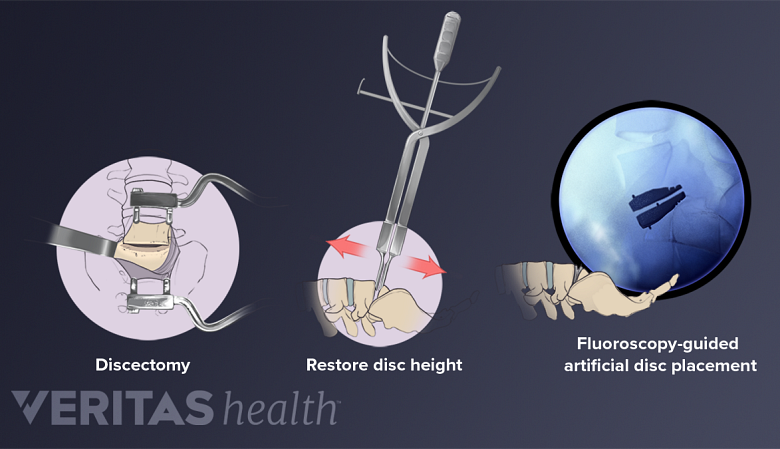
Lumbar artificial disc replacement
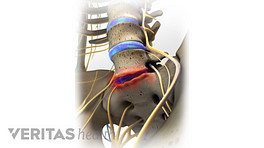
Artificial disc replacement surgery replaces a damaged disc with an artificial one.
In this procedure, the symptomatic lumbar disc is removed and then replaced with an artificial disc designed to maintain the natural movement in the spine. A lumbar artificial disc replacement seeks to minimize pain by reducing instability and muscle tension caused by painful micro-motions at the degenerated disc level.
See Artificial Disc Replacement or Spinal Fusion: Which is Better for You?
Each approach has variations in implants and technique, each has unique advantages and disadvantages, and similar rates of complications have been found to occur from both surgeries. 1 Formica M, Divano S, Cavagnaro L, et al. Lumbar total disc arthroplasty: outdated surgery or here to stay procedure? A systematic review of current literature. J Orthop Traumatol. 2017;18(3):197-215. For example, spinal fusion may lead to future problems in other areas of the spine (called adjacent segment disease). Artificial disc replacement poses the risk of the device failing to maintain motion and a failure to reduce pain.
Data from FDA studies have shown that an artificial disc maintaining motion in the spine decreases the chances of adjacent segment disease. Additionally, study data have shown that artificial disc replacement requires a relatively short recovery time and allows patients to return to activity soon after surgery. 2 Garcia R, Yue JJ, Blumenthal S, et al. Lumbar Total Disc Replacement for Discogenic Low Back Pain: Two-year Outcomes of the activL Multicenter Randomized Controlled IDE Clinical Trial. Spine. 2015;40(24):1873-81.
Proponents of spinal fusion suggest that newer, less invasive surgical techniques and implants can lead to improved outcomes as well as shorter recovery periods following surgery.
There is no single best approach; patients are advised to research their options and discuss the pros and cons of each treatment option with their treating physician.
- 1 Formica M, Divano S, Cavagnaro L, et al. Lumbar total disc arthroplasty: outdated surgery or here to stay procedure? A systematic review of current literature. J Orthop Traumatol. 2017;18(3):197-215.
- 2 Garcia R, Yue JJ, Blumenthal S, et al. Lumbar Total Disc Replacement for Discogenic Low Back Pain: Two-year Outcomes of the activL Multicenter Randomized Controlled IDE Clinical Trial. Spine. 2015;40(24):1873-81.