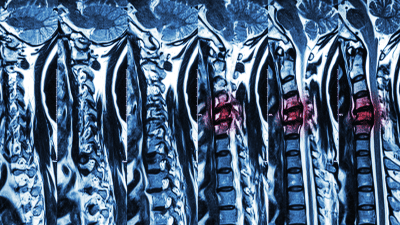
Magnetic resonance imaging (MRI) is a powerful diagnostic tool used to evaluate spine pathology.
For many people, sleeping on your back is one of the best ways to reduce pressure on the spine and promote overnight healing.
Learn about 6 options to help manage your back pain – any one of these could change the way you think about your treatment options.
Walk through 5 common (and costly) mistakes that can delay healing, increase pain, or even lead to long-term damage.
Here are 5 points you need to know about the type of pain at the L4-L5 motion segment.
These 7 healing insights reveal what truly works—and what doesn’t—when it comes to recovering from spinal disc pain and minimizing the chance of a relapse.
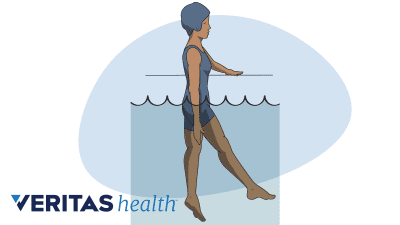
Water therapy or aquatic therapy is an excellent choice for anyone who has too much back pain and/or joint pain for land-based physical therapy.
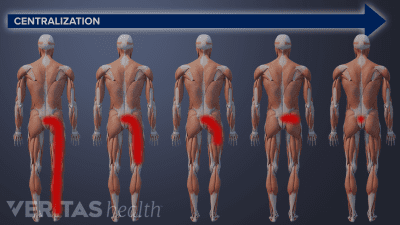
McKenzie therapy is not advised for treating pain from serious back or neck injuries, after spine surgery, or in cases where the pain does not centralize.
You may experience significant pain relief after just one session of McKenzie exercises, but it takes at least 1 to 3 weeks of therapy to get sustained pain relief.
How to relieve SI joint pain while sitting or sleeping? Learn essential tips on ergonomic adjustments, posture, and sleeping position changes to help reduce SI joint pain while sitting or sleeping.