Allograft bone used in spinal fusion is sourced from either living or deceased donors and subsequently undergoes processing to ensure sterility.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
Allografts are easily accessible and are available in diverse forms with favorable handling characteristics. When compared to autografts (the patient’s own bone), allografts possess some attributes that promote bone healing and remodeling and eliminate the need for supplementary surgery and its related complications to harvest bone tissue.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
In This Article:
- Bone Graft for Spine Fusion
- Autograft: The Patient's Own Bone
- Allograft: Bone Graft from a Donor
- Bone Graft Substitutes
How Allografts Help in Spinal Fusion

Allografts promote bone fusion and eliminate the need for additional surgery.
Ideally, a bone graft must contain cells, growth factors, and chemicals to induce bone formation and provide a structural scaffolding where the bone-forming cells can lay down new bone.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- Allografts provide the scaffolding, possess small amounts of growth factors and chemicals, and contain no live cells that induce bone formation.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- The live cells in the allograft are destroyed when the graft undergoes processing treatments to make it sterile for transfusion.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- To overcome the lack of live cells, allografts are augmented with viable cells, tissues, or matrices.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
This process enhances their suitability for achieving successful spinal fusion.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
Availability of Allografts and Their Sources
Allografts can be sourced from living or deceased donors and undergo vigorous treatments to make them suitable for use.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
This table provides an overview of the description and characteristics of various categories of allografts.
| Form of Allograft1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936. | Source1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936. |
|---|---|
| Fresh allograft |
|
| Fresh-frozen allograft |
|
| Freeze-dried allograft |
|
| Demineralized bone matrix |
|
Allografts are sterilized using gamma radiation to prevent the risk of disease transmission from the graft to the host.2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.
Types of Allografts
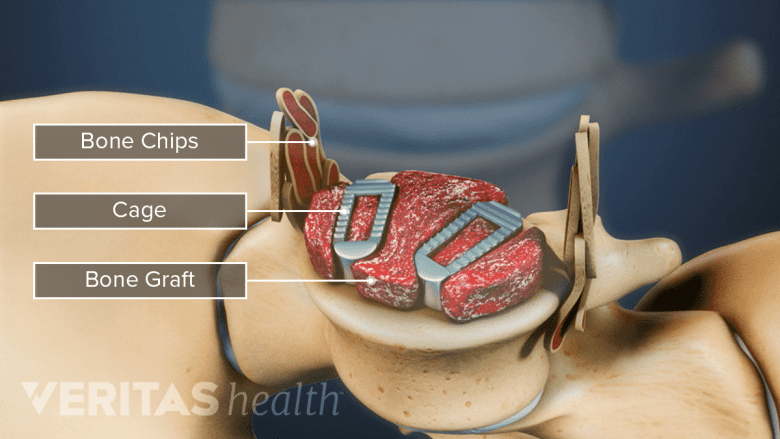
Allograft may be used as a whole piece of bone or bone chips.
Similar to autografts, allografts are categorized as either cortical or cancellous.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- Cortical allografts are dense and have low porosity, making them suitable for use as mechanical strut grafts. They can effectively bear the mechanical loads in the fused segment but have a lower capacity to initiate the bone healing and remodeling process.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- Cancellous allografts have high porosity and less density and function more efficiently as a scaffold to promote bone healing and remodeling.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
The bone may be prepared in 2 ways: as whole pieces or bone chips. Whole pieces include rings of bone taken from the top part of the thigh bone. These pieces are used in interbody spinal fusions, such as ALIF and PLIF. Bone chips are crushed pieces of bone. These chips are used to fill gaps in surgeries like posterolateral gutter spine fusion.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
Demineralized bone matrices
Demineralized bone matrices are prepared from allografts by dissolving the hard (mineralized) tissue of the bone and preserving the soft collagenous and protein extracts and growth factors. These matrices are available as putties, pastes, and injectable gels.2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.
Demineralized bone matrices confer osteoconductive properties (the scaffolding base) and osteoinductive properties (bone repair and remodeling through viable cells).2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429. These allografts are also called viable cellular allografts or cellular bone matrices.
In spinal fusions, demineralized bone matrices are typically used along with another graft tissue (autograft or allograft) and may not be suitable when used independently.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
Allograft Use in Spinal Fusion Surgeries
Allografts are widely used in neck (cervical), upper back (thoracic), and lower back (lumbar) fusions.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
- Successful outcomes have been reported in over 90% of cervical fusions when allografts are used.
- The success rates of lumbar fusions when approached from the back, such as PLIF, are also high when allografts are used.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
- The success rate for lumbar fusion with allograft is lower when the fusion is approached from the side, such as XLIF.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
Research suggests that the best use of allografts is in adolescent patients for the surgical treatment of scoliosis.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
Potential Benefits of Allografts

Allografts are associated with fewer pain complications and infections compared to autografts.
Allografts serve as a viable alternative to autografts and provide the following potential benefits1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.:
- Reduced donor site pain. Allografts eliminate the need for harvesting bone from the patient's own body, minimizing the need for additional surgery.
- Faster procedure. Allografts are readily available, leading to quicker surgical procedures compared to autografts.
- Decreased morbidity. Avoiding a second surgical site for bone extraction lowers the risk of pain, complications, and infections.
- Tailored graft size. Allografts can be prepared in different sizes, accommodating various spinal fusion needs.
Advancements in the safety, quality, and accessibility of allogeneic materials have led to a significant increase in allograft utilization.
Potential Risks of Allografts
While medical professionals continue to harness the potential of allografts, there are a few drawbacks associated with these grafts:
- Lower chance of fusion. Because allograft bone does not contain living bone cells, it is not as effective at stimulating fusion as the patient’s own bone.
- Disease transmission. Allografts pose a risk of disease transmission, such as human immunodeficiency virus and hepatitis, from the donor to the host.2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.,3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448. This risk is higher in fresh-frozen allografts and lesser in freeze-dried allografts.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448. Following proper procurement and sterilization processes eliminates this risk to a large extent.1Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- Graft rejection. In rare cases, the host’s body may trigger an immunogenic reaction, where it identifies the graft as a foreign body and rejects it.2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.,3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
- High cost. While allografts are widely available, the cost of these grafts is higher compared to autografts.3Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.
Through careful donor screening, meticulous processing, and ongoing research, the safety, quality, and availability of allograft materials have substantially improved, fostering a surge in their usage.2D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.
- 1 Cohen JD, Kanim LE, Tronits AJ, Bae HW. Allografts and Spinal Fusion. Int J Spine Surg. 2021 Apr;15(s1):68-93. doi: 10.14444/8056. Epub 2021 Apr 21. PMID: 34376497; PMCID: PMC8092936.
- 2 D'Souza M, Macdonald NA, Gendreau JL, Duddleston PJ, Feng AY, Ho AL. Graft Materials and Biologics for Spinal Interbody Fusion. Biomedicines. 2019 Sep 26;7(4):75. doi: 10.3390/biomedicines7040075. PMID: 31561556; PMCID: PMC6966429.
- 3 Carlisle ER, Fischgrund JS. Bone graft and fusion enhancement. In: Surgical Management of Spinal Deformities. Elsevier; 2009:433-448.

