Featured Health Topics
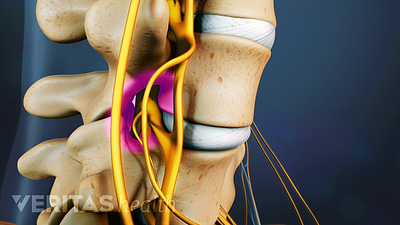
What Is Spinal Stenosis?
Different types of spinal stenosis, including lumbar stenosis and cervical stenosis, have their own pain and symptom profiles.
Exercise and Back Pain
Exercise can help alleviate and prevent back pain by strengthening core muscles, improving flexibility, and promoting overall spine health.
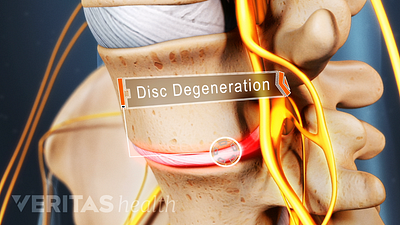
Cervical Degenerative Disc Disease
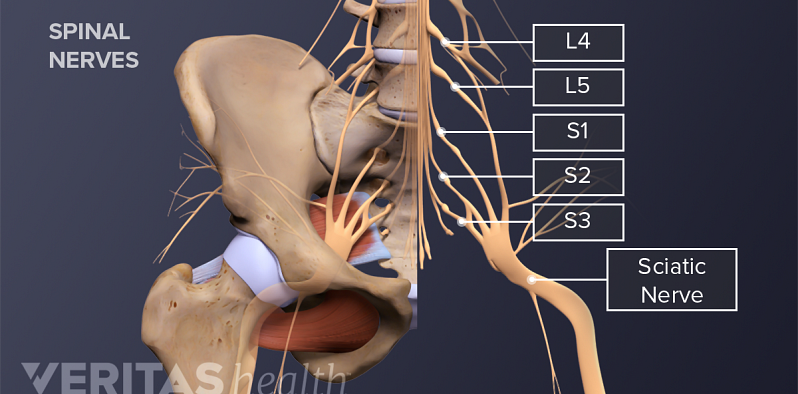
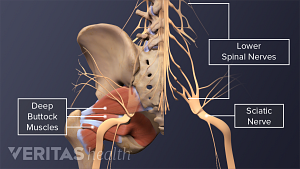
What You Need to Know About Sciatica
Spotlight on your health
Lower Back Pain Causes and Symptoms Video
Chronic pain in the lower back may be caused by the discs or the joints of the spine.
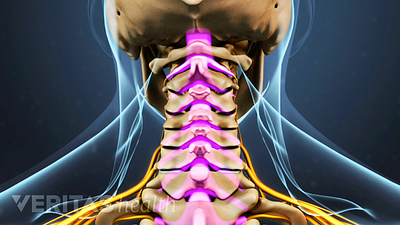
Cervical Spinal Stenosis Video
Learn about cervical stenosis and its symptoms, causes and treatment options in this interactive cervical spinal stenosis video.
Sciatica Causes and Symptoms Video
Sciatica is leg pain caused by a problem in the low back. Watch an animated video that details the causes and symptoms of sciatica.
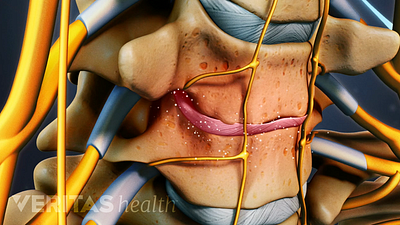
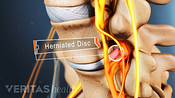
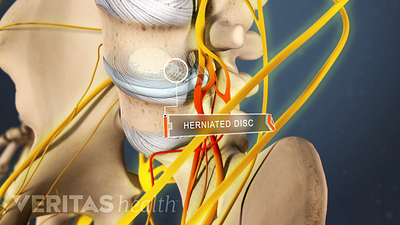
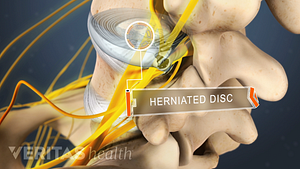
Lumbar Herniated Disc Video
A herniated disc in the lumbar spine can put pressure on spinal nerve roots, causing pain in the lower back or legs.
Editors Top Picks
A complete guide to the causes of buttock muscle pain, sciatica pain in the buttock, and treatments to help relieve pain in the buttock area.
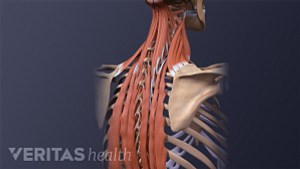
Pain in the shoulder blade region is fairly common and can be quite painful and limiting. It can have many causes, ranging from a muscle strain to a more serious condition that requires immediate medical attention.
Avoid sciatica pain while walking by shortening your stride and engaging your core muscles. Additionally, tucking the tummy in provides extra stability to the spine.
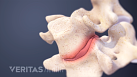
Home remedies that treat inflammation and reduce pressure on the spinal discs provide pain relief and facilitate healing from a herniated disc in the lower back.