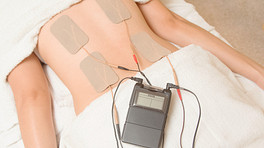
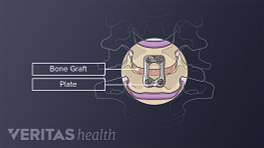
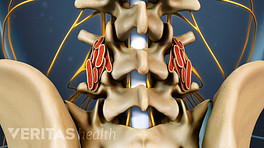
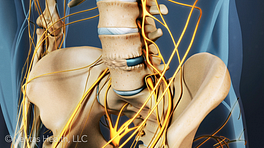
External bone growth stimulation devices are worn outside the skin and do not require surgical implantation or extraction. Typically, the device is worn after spine fusion either as:
- Small, thin skin pads/electrodes that are placed directly over the fusion site and deliver a type of electrical stimulation called Capacitive Coupling (CC), or;
- One or two treatment coil(s) placed into a brace or directly onto the skin that deliver a type of electromagnetic field called a Pulsed ElectroMagnetic Field (PEMF), or a Combined Magnetic Field (CMF).
Unlike an internal (implanted) bone growth stimulator, an external bone growth stimulator may also be prescribed for the patient to use several weeks or months after the fusion surgery if the bone is not fusing as desired.
Depending on the device and the patient's situation, an external bone growth stimulator will be prescribed to be worn for a specific number of hours each day (typically within the range of 2 hours to 9 hours per day). Sometimes the patient may be allowed to break it up into several one- or two-hour sessions each day, or to vary the times that the device is worn each day, to better suit the patient’s schedule. Typically, the external bone growth stimulator will be worn for a period of 3 to 9 months following the surgery.
In This Article:
- How an Electrical Bone Growth Stimulator Helps with Spine Fusion
- Internal Bone Growth Stimulators for Spine Fusion
- External Bone Growth Stimulators for Spine Fusion
An external electrical stimulator is usually lightweight and powered by a battery, so it is very portable. Patients may move about and complete their daily activities as they normally would with the device. However, the surgeon may restrict the patient's activities due to the fusion surgery, and there are some activities that the patient should remove the device for (e.g. swimming, bathing).
See Postoperative Care for Spinal Fusion Surgery
As with the internal stimulators, the external device is not painful and the patient cannot feel any electrical shocks or vibrations while wearing it.
While external electrical stimulation devices are considered very safe, it is important to note that the electromagnetic effects of this type of treatment are unknown for pregnant women and for some types of pacemakers and defibrillators.
Other Considerations for Bone Growth Stimulator and Spine Fusion
A theoretical disadvantage of an external stimulator versus the internal stimulator is that there may be less patient compliance with wearing the external stimulator the required number of hours. Obviously, if the patient does not wear the device, the benefits from the treatment will not be realized.
A potential advantage of external stimulators versus internal stimulators is that the external device is usually a less expensive treatment option and does not require the potential second surgery to remove the battery pack.
Another major advantage is that an external bone stimulator can be added after the fusion has been done if there is concern that the bone graft is not healing and a nonunion is developing.