Magnetic resonance imaging (MRI) is used when high-resolution images of the body’s tissues are required for diagnosis and treatment.
For back or neck pain, MRI scans are a preferred diagnostic tool for investigating musculoskeletal problems with or without neurologic deficits when previous imaging is suggestive of spinal pathology. However, the presence of certain implants or prostheses may interfere with the MRI process, making it a health risk.
In This Article:
- MRI Scan of the Spine
- Indications and Contraindications for an MRI Scan
Indications for a Spinal MRI Scan
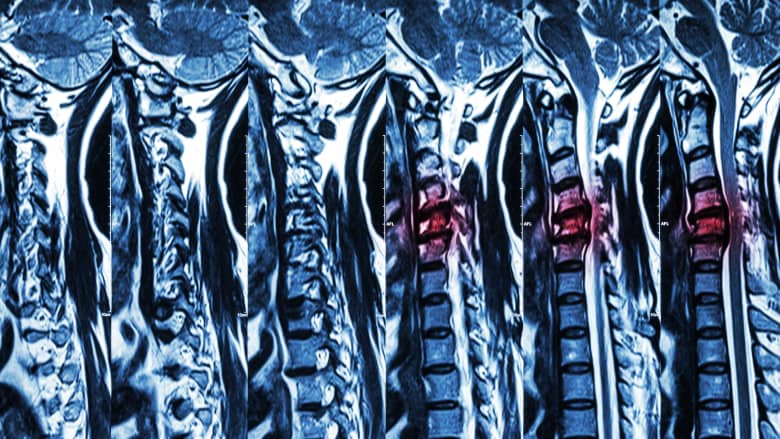
Severe spinal cord compression is evaluated in detail on an MRI scan.
An MRI scan may be used to diagnose, further investigate, and/or plan the treatment of neck pain or back pain that fulfills one or more of the following criteria:
- Medical emergencies. A confirmed or suspected medical emergency, such as spinal cord compression, which requires immediate surgical intervention.
- Developing infections or tumors. Back or neck pain is accompanied by constitutional symptoms (such as loss of appetite, weight loss, fever, chills, shakes, or night pain) that may indicate the presence of a tumor or infection.
- Radiculopathies. Back or neck pain is caused by suspected spinal nerve compression (radiculopathy) due to conditions such as spinal stenosis or herniated disc(s).
- Recurrent or chronic symptoms. There is an incomplete resolution of pain after a series of first-line treatments, such as pain-relieving medications and physical therapy, have been tried for several weeks —and the cause of pain needs further investigation.
- Progressively worsening symptoms. The pain and related symptoms such as numbness and weakness have progressively worsened despite nonsurgical treatment.
- Chronicity of fractures. An MRI is not the best diagnostic tool to examine problems in the bones, such as vertebral fractures. However, an MRI scan can detect swelling in the soft bone marrow tissue inside the bone, providing insight into whether a fracture is acute, chronic, benign, or malignant.1Kumar Y, Hayashi D. Role of magnetic resonance imaging in acute spinal trauma: a pictorial review. BMC Musculoskelet Disord. 2016;17:310. Published 2016 Jul 22. doi:10.1186/s12891-016-1169-6
MRI scans may also be used to view postsurgical healing of tissues.
Contraindications for a Spinal MRI Scan

An MRI scan is contraindicated for individuals with a spinal cord stimulator.
The presence of any ferromagnetic material in the body is a major contraindication for an MRI scan.2Hackenbroch C, Wafa M, Klinger S, Mauer UM. Magnetic resonance imaging in the presence of projectiles and projectile fragments: Artefacts, image quality, rotation and movement. Magn Reson Imaging. 2019;57:143-150. doi:10.1016/j.mri.2018.11.019 These materials (discussed below) are strongly attracted to magnets and may get dislodged or overheated, causing serious health risks to the patient.
Before an MRI scan, all external metallic items should be removed. Such items may include external hearing aids, jewelry, watches, and clothing with zippers, metallic threads, or metal buttons and fasteners.
Due to their metallic nature and magnetic properties, a spinal MRI scan may not be performed when one or more of the following devices are implanted in the body.
- Cardiac pacemakers or implantable cardioverter defibrillator (ICD) devices, especially older models, can be affected by the MRI scanner’s magnets, causing the devices to move, heat up, or deactivate. Electromagnetic and radiofrequency signals may also cause older devices to inappropriately defibrillate or alter pacing.3Markman TM, Halperin HR, Nazarian S. Update on MRI Safety in Patients with Cardiac Implantable Electronic Devices. Radiology 2018; 00:1-2. July 3, 2018. doi:10.1148/radiol.2018180360
- Spinal cord stimulators (SCS) implanted under the skin may become dislodged or damaged by the MRI machine’s magnet.
- Cochlear implants that assist hard-of-hearing or deaf people can be damaged or displaced during the MRI procedure. A damaged or displaced implant can lead to discomfort, significant pain, or device malfunction. The implant may also interfere with the quality of the scan.4Kim BG, Kim JW, Park JJ, Sung Huhn Kim SH, Kim HN, Choi JY. Adverse Events and Discomfort During Magnetic Resonance Imaging in Cochlear Implant Recipients. JAMA Otolaryngol Head Neck Surg. 2015;141(1):45-52. doi:10.1001/jamaoto.2014.2926
- Dental materials, such as braces, retainers, fixed appliances, metallic fillings, or implants may become heated or displaced during an MRI scan. Dental implants and crowns may also affect the quality of the image.5Chockattu SJ, Suryakant DB, Thakur S. Unwanted effects due to interactions between dental materials and magnetic resonance imaging: a review of the literature. Restor Dent Endod. 2018;43(4):e39. Published 2018 Aug 30. doi:10.5395/rde.2018.43.e39
- Aneurysm clips or coils may become dislodged from the blood vessel and cause significant bleeding.
- Shrapnel or metal shavings in the body, such as from working in construction or the military, may move or become heated during an MRI, causing pain, bleeding, and potentially more serious complications.
While most of these implants are considered unsafe for magnetic resonance imaging, some may be safe when magnetic resonance scanners with weaker field strength are used.6Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/
It is important to discuss the risks posed by devices or implants with the treating physician or MRI technician. It is also advised to bring information about the specific make and model of any implanted device to the appointment so that any risk to patient safety can be determined. If there is any doubt about the safety of an MRI, other imaging techniques, such as CT scan, may be used.
Contraindications for Spinal MRI with Contrast
The MRI contrast agent, gadolinium, may cause potential complications in some patients with kidney and/or heart problems and may be contraindicated in such cases. These contraindications include but are not limited to6Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/:
- Patients on dialysis
- Patients with a history of kidney disease (renal transplant, single kidney, renal cancer)
- Patients with impaired kidney function may develop nephrogenic systemic fibrosis (NSF), a serious illness caused by the improper breakdown of the contrast agent in the kidney
- Patients with diabetes mellitus or hypertension who are receiving treatment with medications
In some cases, blood work may be ordered to help analyze the kidney function and determine if it can metabolize and filter gadolinium if the agent is injected into the body.7Weinreb JC, Rodby RA, Yee J, et al. Use of intravenous gadolinium-based contrast media in patients with kidney disease: consensus statements from the american college of radiology and the national kidney foundation. Radiology. 2021;298(1):28-35. doi:10.1148/radiol.2020202903,8Mathur M, Jones JR, Weinreb JC. Gadolinium deposition and nephrogenic systemic fibrosis: a radiologist’s primer. RadioGraphics. 2020;40(1):153-162. doi:10.1148/rg.2020190110
A known allergy to gadolinium or having received a dose of the contrast within the last 24 hours are also contraindications for its use.6Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/
MRI of Spine Fusion Instrumentation
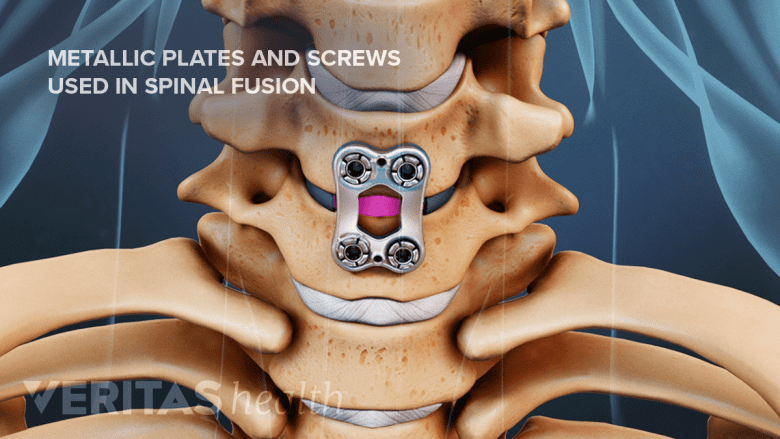
Individuals with spinal implants like screws or cages may have supervised MRI scans.
Patients who have had metallic devices surgically placed in their spine, such as pedicle screws or spinal interbody cages, may have an MRI scan under supervision, but the resolution of the scan is often severely obscured by the metal device and the quality of the imaging study is poor.9Knott PT, Mardjetko SM, Kim RH, et al. A comparison of magnetic and radiographic imaging artifact after using three types of metal rods: stainless steel, titanium, and vitallium. Spine J. 2010;10(9):789-794. doi:10.1016/j.spinee.2010.06.006
In general, stainless-steel implants cause more severe artifacts than titanium. Titanium is a paramagnetic material that is generally not affected by the magnetic field of MRI.10Kim YH, Choi M, Kim JW. Are titanium implants actually safe for magnetic resonance imaging examinations?. Arch Plast Surg. 2019;46(1):96-97. For this reason, titanium has gained popularity for spine fusion instrumentation, and stainless steel has largely fallen out of favor due to the inability to get a clear postsurgical MRI scan.
MRI During Pregnancy and Breastfeeding
Standard MRI is typically considered safe in pregnant patients (all trimesters) although its use is limited and based on the physician’s discretion. MRI with contrast, however, is not used in pregnant patients but may be used with caution in breastfeeding mothers.6Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/,11Ibrahim MA, Hazhirkarzar B, Dublin AB. Gadolinium Magnetic Resonance Imaging. [Updated 2022 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482487/
The FDA determines the MRI contrast agent, gadolinium, as a class C agent, which means that there is no established evidence regarding its safety during pregnancy. A very small fraction of this agent is excreted through breast milk and absorbed by the infant.6Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/ Breastfeeding mothers are advised to pump and store breastmilk before the MRI with contrast scan and may resume breastfeeding 24 to 48 hours after the scan is performed.11Ibrahim MA, Hazhirkarzar B, Dublin AB. Gadolinium Magnetic Resonance Imaging. [Updated 2022 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482487/
MRI is generally safe and produces high-resolution scans for diagnosing and treating spinal conditions. When used in appropriate patients, this diagnostic tool can help understand underlying pathologies and plan treatment more effectively.
- 1 Kumar Y, Hayashi D. Role of magnetic resonance imaging in acute spinal trauma: a pictorial review. BMC Musculoskelet Disord. 2016;17:310. Published 2016 Jul 22. doi:10.1186/s12891-016-1169-6
- 2 Hackenbroch C, Wafa M, Klinger S, Mauer UM. Magnetic resonance imaging in the presence of projectiles and projectile fragments: Artefacts, image quality, rotation and movement. Magn Reson Imaging. 2019;57:143-150. doi:10.1016/j.mri.2018.11.019
- 3 Markman TM, Halperin HR, Nazarian S. Update on MRI Safety in Patients with Cardiac Implantable Electronic Devices. Radiology 2018; 00:1-2. July 3, 2018. doi:10.1148/radiol.2018180360
- 4 Kim BG, Kim JW, Park JJ, Sung Huhn Kim SH, Kim HN, Choi JY. Adverse Events and Discomfort During Magnetic Resonance Imaging in Cochlear Implant Recipients. JAMA Otolaryngol Head Neck Surg. 2015;141(1):45-52. doi:10.1001/jamaoto.2014.2926
- 5 Chockattu SJ, Suryakant DB, Thakur S. Unwanted effects due to interactions between dental materials and magnetic resonance imaging: a review of the literature. Restor Dent Endod. 2018;43(4):e39. Published 2018 Aug 30. doi:10.5395/rde.2018.43.e39
- 6 Ghadimi M, Sapra A. Magnetic Resonance Imaging Contraindications. [Updated 2022 May 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551669/
- 7 Weinreb JC, Rodby RA, Yee J, et al. Use of intravenous gadolinium-based contrast media in patients with kidney disease: consensus statements from the american college of radiology and the national kidney foundation. Radiology. 2021;298(1):28-35. doi:10.1148/radiol.2020202903
- 8 Mathur M, Jones JR, Weinreb JC. Gadolinium deposition and nephrogenic systemic fibrosis: a radiologist’s primer. RadioGraphics. 2020;40(1):153-162. doi:10.1148/rg.2020190110
- 9 Knott PT, Mardjetko SM, Kim RH, et al. A comparison of magnetic and radiographic imaging artifact after using three types of metal rods: stainless steel, titanium, and vitallium. Spine J. 2010;10(9):789-794. doi:10.1016/j.spinee.2010.06.006
- 10 Kim YH, Choi M, Kim JW. Are titanium implants actually safe for magnetic resonance imaging examinations?. Arch Plast Surg. 2019;46(1):96-97.
- 11 Ibrahim MA, Hazhirkarzar B, Dublin AB. Gadolinium Magnetic Resonance Imaging. [Updated 2022 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK482487/

